In September 2020, General Secretary Xi Jinping stated during the 75th United Nations General Assembly that China will increase its national voluntary contributions, implement stronger policies and measures, and strive to peak its carbon dioxide emissions before 2030, aiming for carbon neutrality by 2060 under the "dual carbon" goals. In this context, the proportion of renewable energy sources in the future energy structure will continue to rise, making carbon dioxide resource utilization very important.
In CO₂ reduction reactions, the products are diverse, including common products such as CO, C1 compounds (CH₄, CH₃OH, HCOOH, etc.), C2 compounds (C₂H6, C₂H₅OH, CH₃COOH, etc.), and C2+ hydrocarbons, among others.
In CO₂ reduction reactions, the coexistence of water reduction competitive reactions and the introduction of air components into the detection system can pose challenges in the accurate identification of CO₂ reduction reaction products.
Furthermore, during the catalyst preparation process, the use of solvents, reactants, and surfactants, among other organic substances, may leave carbonaceous residues in the catalyst and may decompose into small molecular products such as CO and CH₄ during the reaction, potentially increasing the product yield in CO₂ reduction reactions. This could lead to a potential for "false" yield and seriously interfere with the assessment of the true activity of the catalyst in the CO₂ reduction reaction.
Therefore, in assessing the performance of catalysts in CO₂ reduction reactions, it is necessary not only to confirm the yield of each reaction product but also to further confirm the real sources of each product. This means verifying that the products are generated from the catalytic conversion of CO₂, not from the decomposition of carbonaceous organic matter contained in the catalyst, in order to effectively measure the true activity of the catalyst in CO₂ reduction reactions.
Currently, existing Gas Chromatography-Mass Spectrometry (GC-MS) techniques for tracing isotopes in CO₂ reduction products lack clear standards for selecting chromatographic columns and testing conditions when tracing different products.
In the practical application of existing GC-MS isotope tracing technology for CO₂ reduction products:
1. Due to the diversity of products and the separation ability of chromatographic columns, inappropriate chromatographic columns can result in impure single peak components in the total ion chromatogram of gas chromatography. In scanning mode, significant interference with product tracing can be observed in the detection spectrum, with many other components, including various ion fragments such as 13C, N+, O+, HO+, H₂O+, present (see Figure 1).
2. When analyzing specific products in ion mode, there is a shortcoming in effective isotope tracing analysis. For example, when m/z=17, it may originate from fragment ions of reactant water (HO+), interfering with the molecular ion peak of methane (13CH₄+). When m/z=29, it may originate from fragment ions of 13CO₂ (13CO+) and natural abundance nitrogen isotope molecules and ions in the air (15N, 14N+), interfering with the molecular ion peak of 13CO (13CO+).
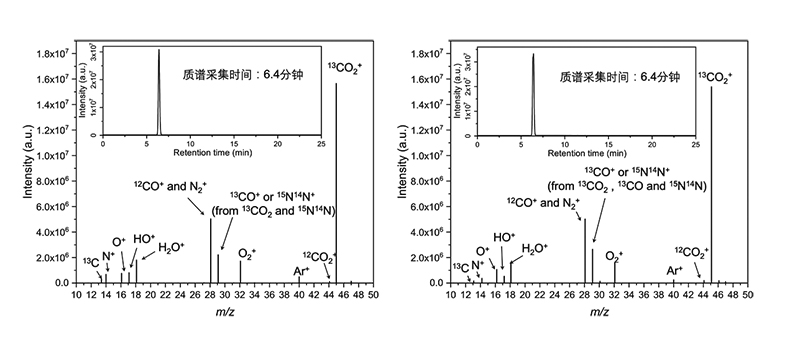
Figure 1. GC-MS spectrum of reactants (13CO₂ and H₂O) detected with a chromatographic column without good separation capability (left);
GC-MS spectrum of reactants (13CO₂ and H₂O) and products (13CO) detected with a chromatographic column without good separation capability (right).
Therefore, due to chromatographic separation issues, common chromatographic columns cannot efficiently separate reactants (such as CO₂ and H₂O) and products (such as CO), and 13CO₂ entering the mass spectrometer can result in peaks attributed to 13CO₂ molecular ions (m/z=45) as well as peaks attributed to 13CO₂ fragment ions (m/z=29, m/z=16, m/z=13). The presence of 13CO₂ fragment ion peaks seriously interferes with the detection of 13CO and other products.
To achieve correct, reliable, and consistent isotope tracing data for CO₂ reduction products, the establishment of detection methods and standards is essential. Perfectlight Technology now offers testing services for tracking the real carbon source in CO₂ reduction experiments.
We focus on different types of CO₂ reduction reactions, such as photocatalysis/photothermal catalysis/photoelectrocatalysis/electrocatalysis, and for different products, by selecting and optimizing the type of chromatographic columns and testing conditions, we establish isotope separation analysis methods and obtain standard mass spectra. We are creating new technologies and clear standards for tracing isotopically labeled products in CO₂ reduction reactions, efficiently separating substances that interfere with product tracing analysis, ensuring that each chromatographic peak in the total ion chromatogram is a pure peak of a single species, and enabling effective tracing of different products in CO₂ reduction reactions. This allows for the accurate measurement of the true activity of catalysts in CO₂ reduction reactions.
Perfectlight Technology offers 13C, 18O, and D₂O isotope tracing qualitative and quantitative testing for different products in CO₂ reduction reactions, including photocatalysis, photothermal catalysis, photoelectrocatalysis, and electrocatalysis, among others. We have a professional testing team and provide comprehensive spectrum analysis services.
Photocatalysis, Photothermal Catalysis, Photoelectrocatalysis, Electrocatalysis
Carbon Monoxide, Hydrocarbons, Aldehydes, Alcohols, Acids
13C, 18O, and D₂O
Labsolar-6A All-Glass Automatic Online Trace Gas Analysis System + Imported Chromatograph
This testing service establishes isotope separation analysis methods by selecting and optimizing chromatographic column types and testing conditions, obtaining standard mass spectra, and creating new technologies and clear standards for tracing isotopically labeled products in CO₂ reduction reactions. It efficiently separates substances that interfere with product tracing analysis, ensuring that each chromatographic peak in the total ion chromatogram is a pure peak of a single species. This enables effective tracing of different products in CO₂ reduction reactions and allows for the accurate measurement of the true activity of catalysts in CO₂ reduction reactions.
"CO₂ Reduction Experiment 13C Labeling Test Request Form" is available for download. Please log in to view!