Photocatalysis is the combination of photocatalysis and electrocatalysis, which maximizes the advantages of both technologies.
Compared to photocatalysis, in photocatalytic reactions, the photoelectrode can utilize sunlight to generate photoexcited electrons, enhancing reaction activity and catalytic efficiency.
Compared to electrocatalysis, photocatalytic reactions significantly reduce the injection of external energy, effectively reducing energy consumption and environmental pollution.
Photocatalysis can also make catalysts that are unsuitable for photocatalysis due to band structure mismatch applicable to photocatalysis under appropriate external voltage conditions.
Photocatalytic reactions can be classified into various types based on reactants and reaction types, including photocatalytic water splitting for hydrogen production, photocatalytic degradation, photocatalytic CO2 reduction, photocatalytic nitrogen reduction for ammonia synthesis, and more.
The photocatalytic reaction process is an electrochemical process under the influence of light, involving the absorption of light that puts electrons in an excited state, leading to charge transfer. Due to the existence of a band gap in semiconductor materials, the interaction between valence band electrons and conduction band electrons is weak. After being excited by light, valence band electrons in the semiconductor move to the conduction band, leaving behind holes, initiating photoelectrochemical reactions.
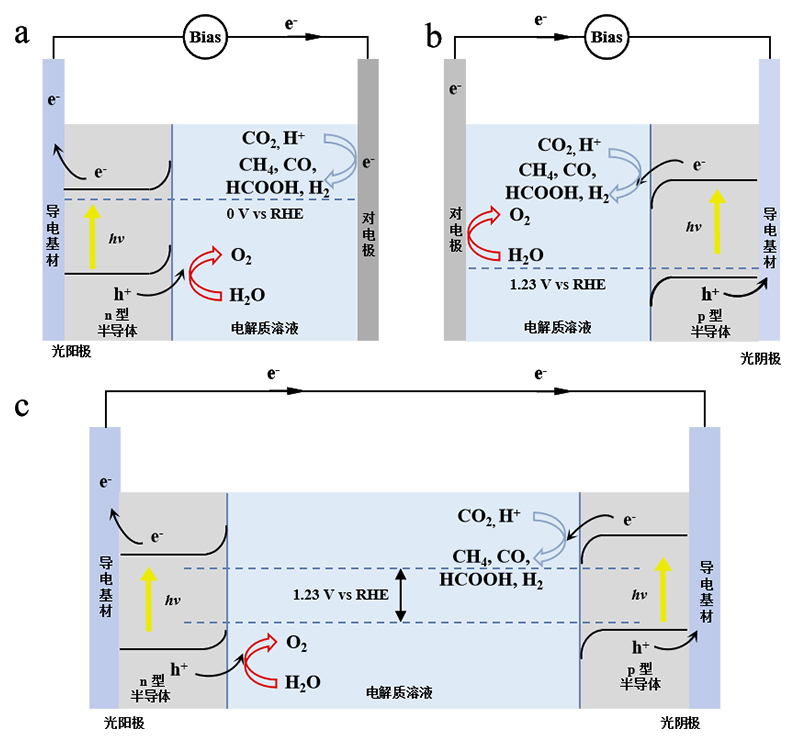
Figure 1. Three common photocatalytic reaction cells: (a) Photoanode photocatalytic reaction cell; (b) Cathode photocatalytic reaction cell; (c) Series-connected photocatalytic reaction cell

Photocatalysis is the combination of photocatalysis and electrocatalysis, which maximizes the advantages of both technologies.
Semiconductor-based photocatalytic reaction cells mainly consist of four parts:
1. Photoelectrode: Also known as the working electrode, it is the core component in the photocatalytic reaction cell, composed of semiconductor materials for capturing light energy.
In general, the n-type semiconductor thin film/conductive substrate is called the photoanode, and the p-type semiconductor thin film/conductive substrate is called the cathode. Common preparation methods for photoelectrodes involve attaching semiconductor materials to transparent conductive oxide-coated glass with low work functions, such as indium tin oxide (ITO) or fluorine-doped tin oxide (FTO), and using photoelectrodes clamped with electrode fixtures as shown in Figure 2(a).
2. Counter Electrode: When studying single photoanodes or photocathodes, a counter electrode is introduced to serve as the other half of the electrochemical reaction site. The counter electrode is usually made of a material with good charge transfer performance and low reaction overpotential, typically Pt electrodes as shown in Figure 2(b).
3. Reference Electrode: To ensure precise and stable potential applied to the photoelectrode, a reference electrode is often installed, creating a three-electrode system for the entire photocatalytic reaction cell. Common choices for reference electrodes include Ag/AgCl electrodes, Hg/Hg₂Cl₂ electrodes, or Hg/HgO electrodes as shown in Figure 2(c).
4. Suitable Electrolyte Solution: The electrolyte solution in the photocatalytic reaction cell serves the purpose of connecting the circuit and needs to have good ionic conductivity.
Depending on the nature of the photoelectrode material in photocatalytic reactions, the pH value and composition of the electrolyte solution may vary. When choosing an electrolyte solution, it is important to ensure that it does not react with the selected reference electrode.
Electrolyte solutions can be divided into three types:
1) Acidic Electrolyte Solutions: Such as H₂SO₄ solution;
2) Neutral Electrolyte Solutions: Such as Na₂SO₄ solution, K₂SO₄ solution, etc.;
3) Alkaline Electrolyte Solutions: Such as NaOH solution, KOH solution, etc.

Figure 2. (a) Electrode fixture; (b) Pt electrode; (c) Hg/Hg₂Cl₂ electrode; (d) Ag/AgCl electrode