一、Basic Principles of Gas Chromatograph:
Chromatography, also known as chromatographic analysis or chromatography, is a physical and chemical separation method that utilizes the characteristics of substance solubility and adsorption. The separation principle is based on the differences in the interactions of various components in the mixture between the mobile phase and the stationary phase.
Gas chromatography (GC), which uses a gas as the mobile phase, is a highly mechanized chromatographic method widely used for the quantitative analysis of low molecular weight complex substances.
Mobile Phase: The fluid that carries the sample through the entire system, also known as the carrier gas.
Stationary Phase: The immobile phase, which includes the support material, stationary liquid, or packing material in the chromatographic column.
二、Components of a Gas Chromatograph:
A gas chromatograph is mainly composed of a gas delivery system, injection system, separation system, detection and temperature control system, and recording system.
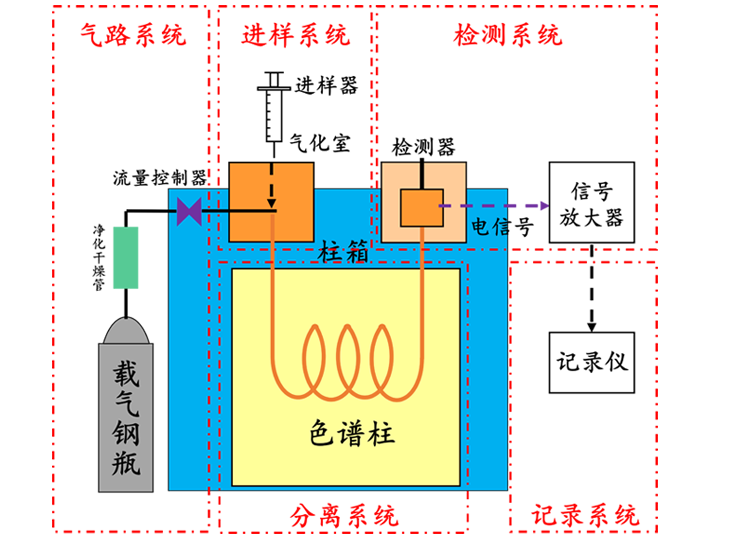
Figure 1. Schematic Structure of a Gas Chromatograph
1. Gas Delivery System of a Gas Chromatograph
The gas delivery system of a gas chromatograph includes a gas source, purification and drying column, and carrier gas flow control device. It is a closed gas path system where the carrier gas continuously flows. The gas tightness, accuracy of flow rate monitoring, and stability of carrier gas flow rate in the gas delivery system of the gas chromatograph are all important factors affecting the performance of the gas chromatograph.
Commonly used carrier gases in gas chromatography include hydrogen, nitrogen, and argon, with a purity requirement of above 99.999%. They should be chemically inert and not react with the components being tested. Carrier gas selection not only considers the separation effect of the components but also takes into account the detector's sensitivity under different carrier gas conditions.
2. Injection System of a Gas Chromatograph
The injection system of a gas chromatograph mainly includes an injector and a vaporization chamber.
(1) Injector: Different injectors are used depending on the phase of the components to be tested. Liquid samples are typically introduced using a microsyringe, as shown in Figure 2. Gas samples are injected using the gas chromatograph's built-in rotary six-way valve or a microsyringe with a fine needle, as shown in Figure 2.

Figure 2. Gas and Liquid Injectors
Solid samples are usually dissolved in an appropriate solvent and then introduced using a microsyringe in liquid form.
(2) Vaporization Chamber: The vaporization chamber is generally made of stainless steel tubing with an external heating wire. Its role is to completely vaporize the liquid sample into vapor instantly. The vaporization chamber should have a sufficiently large heat capacity and no catalytic effect to ensure the instant vaporization of the sample without decomposition.
3. Separation System of a Gas Chromatograph
The separation system of a gas chromatograph is the core part of the instrument, responsible for separating various components in the sample.
The separation system consists of a column oven, chromatographic column, and temperature control components.
There are mainly two types of chromatographic columns: packed columns and capillary columns. Column materials include metals, glass, fused silica, polytetrafluoroethylene (PTFE), etc.
The separation efficiency of the chromatographic column depends not only on the column length, diameter, and shape but also on factors such as the type of column packing material and operating conditions.
4. Detection System of a Gas Chromatograph
The detection system of a gas chromatograph converts the concentration or mass changes of separated components into electrical signals. After amplification, these signals are recorded by a recorder, producing a chromatogram.
The performance of the detector directly affects the accuracy of the final analytical results of the gas chromatograph.
Based on the detection principle, detectors can be categorized into concentration detectors and mass detectors.
(1) Concentration Detectors: These measure the instantaneous concentration changes of the sample components in the carrier gas, with the detector response signal being proportional to the concentration of the components. Examples include the Thermal Conductivity Detector (TCD) and the Electron Capture Detector (ECD).
(2) Mass Detectors: These measure the rate of entry of the sample components carried by the carrier gas into the detector. The detector response signal is proportional to the mass of the components entering the detector per unit time. Examples include the Flame Ionization Detector (FID) and the Flame Photometric Detector (FPD).
5. Temperature Control System of a Gas Chromatograph
In a gas chromatograph, temperature control is crucial as it directly affects the separation efficiency of the chromatographic column, detector sensitivity, and stability.
The temperature control system primarily targets the vaporization chamber, chromatographic column, and detector.
In the vaporization chamber, it ensures the complete and instantaneous vaporization of the liquid sample. In the column oven, it ensures complete separation of components. For samples with a wide range of components, temperature control in the column oven needs to be programmed to optimize separation conditions for different components.
Gas chromatographs use two temperature control methods: constant temperature and programmed temperature ramping.
(1) Constant Temperature Control: This mode is used for simple samples with a narrow boiling range, as well as for gas and liquid sample analyses.
(2) Programmed Temperature Ramp Control: This method involves stepwise temperature increases within a single analysis cycle. It allows for improved separation efficiency, shorter analysis times, and is especially useful for complex samples with a wide boiling range.
6. Recording System of a Gas Chromatograph
The recording system of a gas chromatograph is primarily used to record the detector's detection signals, perform quantitative data processing and recording. Some gas chromatographs are equipped with computers for automated measurement of peak areas, providing accurate quantitative analysis data and automated post-analysis of chromatographic data.
三、Analysis Process of a Gas Chromatograph
The carrier gas of the gas chromatograph flows out from a high-pressure cylinder, passes through a pressure reducing valve to reach the required pressure for the gas chromatograph, undergoes purification and drying in a purification column, and then passes through a pressure regulator and a flowmeter. The carrier gas, at a stable pressure and constant speed, mixes with the vaporized sample in the vaporization chamber and is transported into the chromatographic column for separation. After separation, each component enters the detector for analysis.
The detector converts the concentration or mass changes of the sample components into electrical signals, which are amplified and recorded on a recorder, generating the chromatographic elution curve. Qualitative analysis can be performed based on the retention time on the chromatographic elution curve, while quantitative analysis can be carried out based on the peak area or peak height. The specific analysis process is shown in Figure 3.
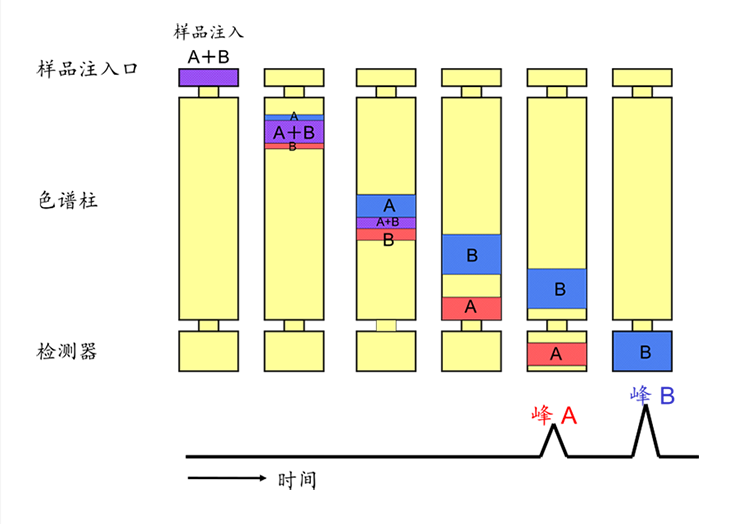
Figure 3. Analysis Process of a Gas Chromatograph